The electron spin number (ms) -describes the spin of the electron either up or down.

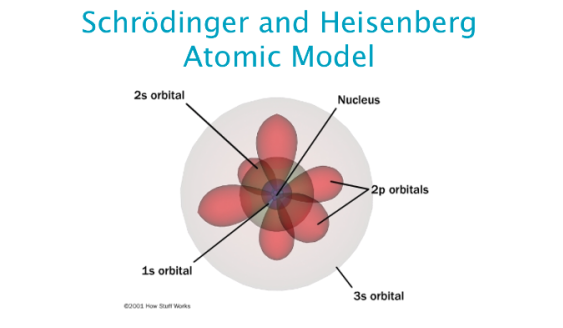
The magnetic quantum number (ml) – describes the energy level in the subshell.The principal quantum number (n) - describes the energy of the electron and its distance from the nucleus.Orbital angular momentum (l) – Describes the shape of the orbital.Each electron has its own unique 4 quantum numbers as according to the Pauli exclusion principle no two electrons can share the same combination of quantum numbers. The quantum numbers of an electron give it its own identity and these combined are described by a wave function that obeys the Schrodinger equation. This expanded the understanding of the electron to have 4 quantum numbers. In the Bohr Model the electron is treated as a particle in fixed orbits around the nucleus where as Schrodinger’s model treated the electron as a wave that occupies three dimensional space. This equation could be used to solve and give an accurate description of the energy of an electron in an atom due to vibrational modes and the creation of peaks and troughs when these electrons are treated as waves.Įrwin Schrodinger was important in the driving of a mathematical and quantum mechanics for developing the understanding of electrons in moving away from the Bohr Model.
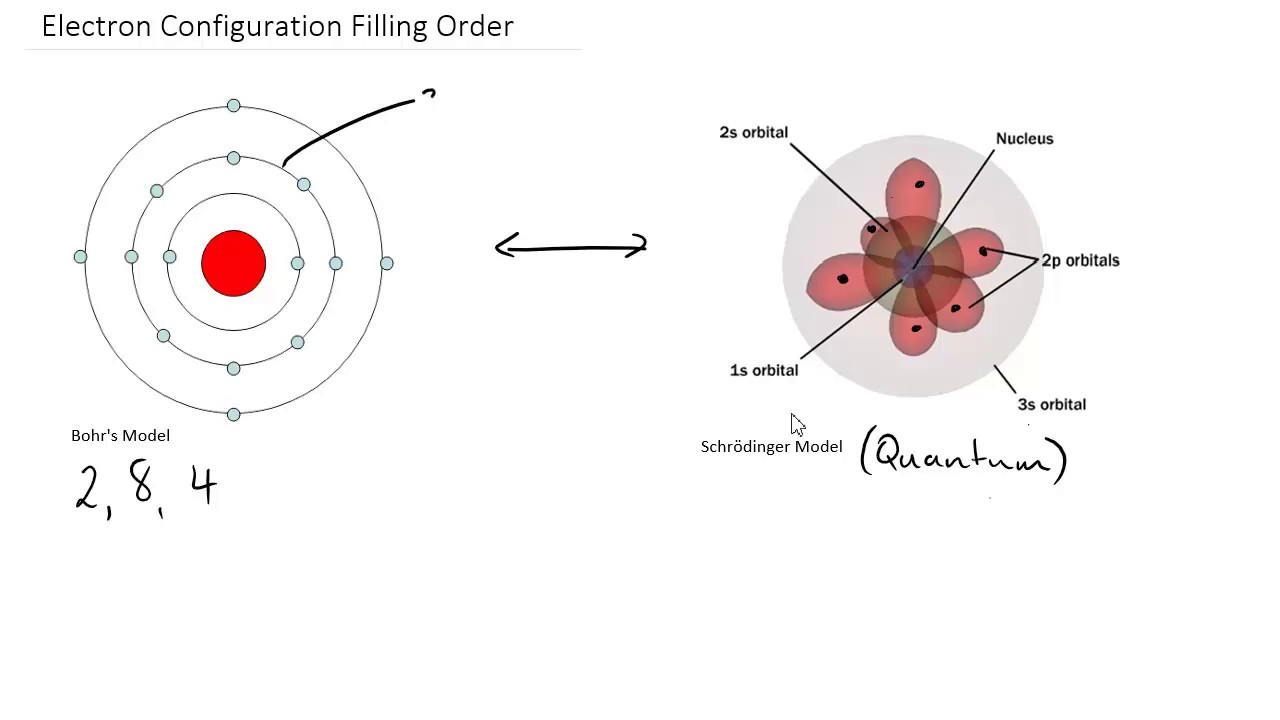
The model of the electron in the nucleus was further developed by the work of Erwin Schrodinger and his development of his wave equation.
